Why Does My Voice Sound Weird with Hearing Aids? Occlusion Explained
The first few days with new hearing aids can bring up something nobody
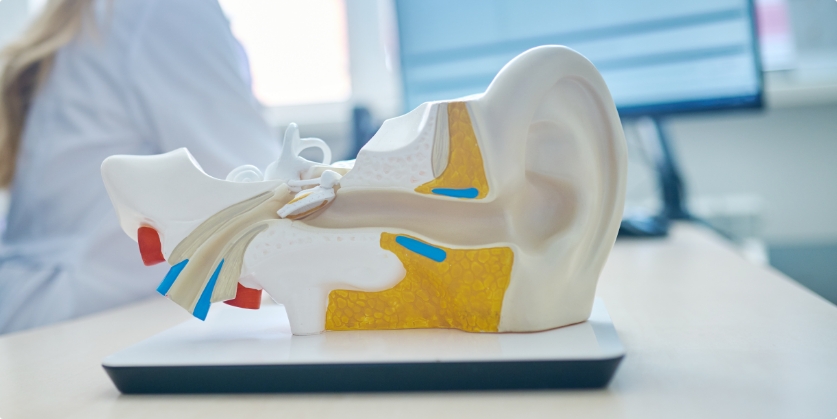

The first few days with new hearing aids can bring up something nobody
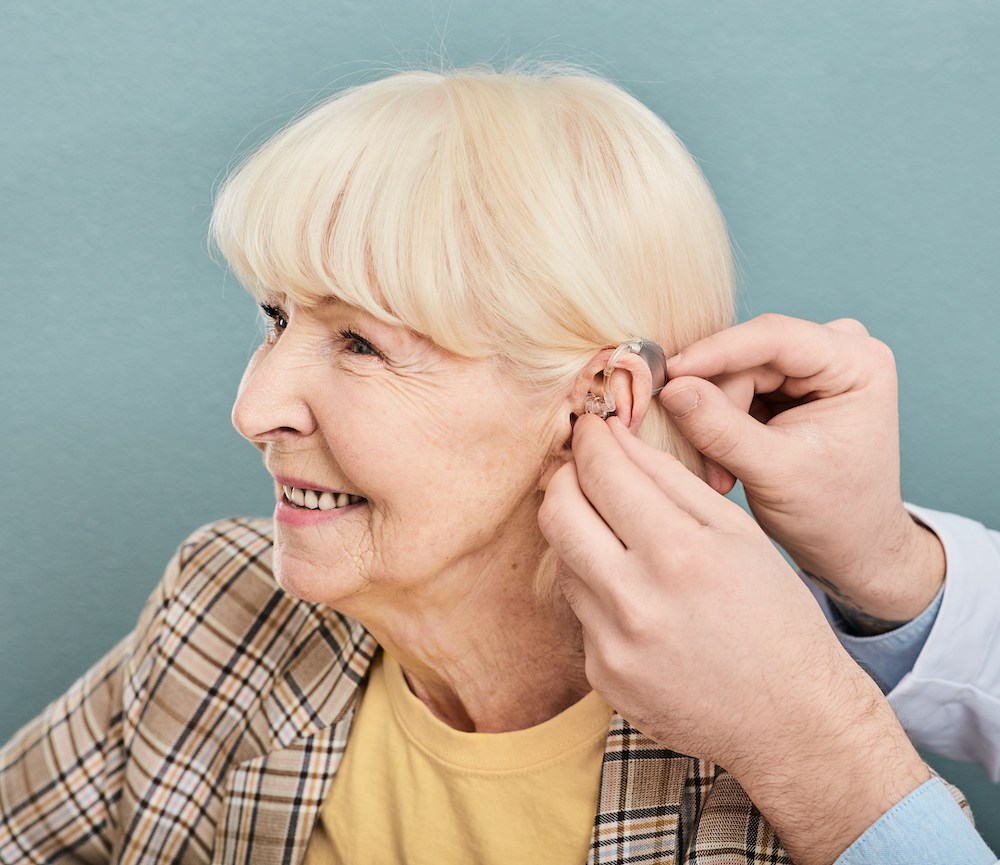
Hearing aid technology has come a long way in recent years, and 2025

Helping children with hearing loss isn’t just about making smaller